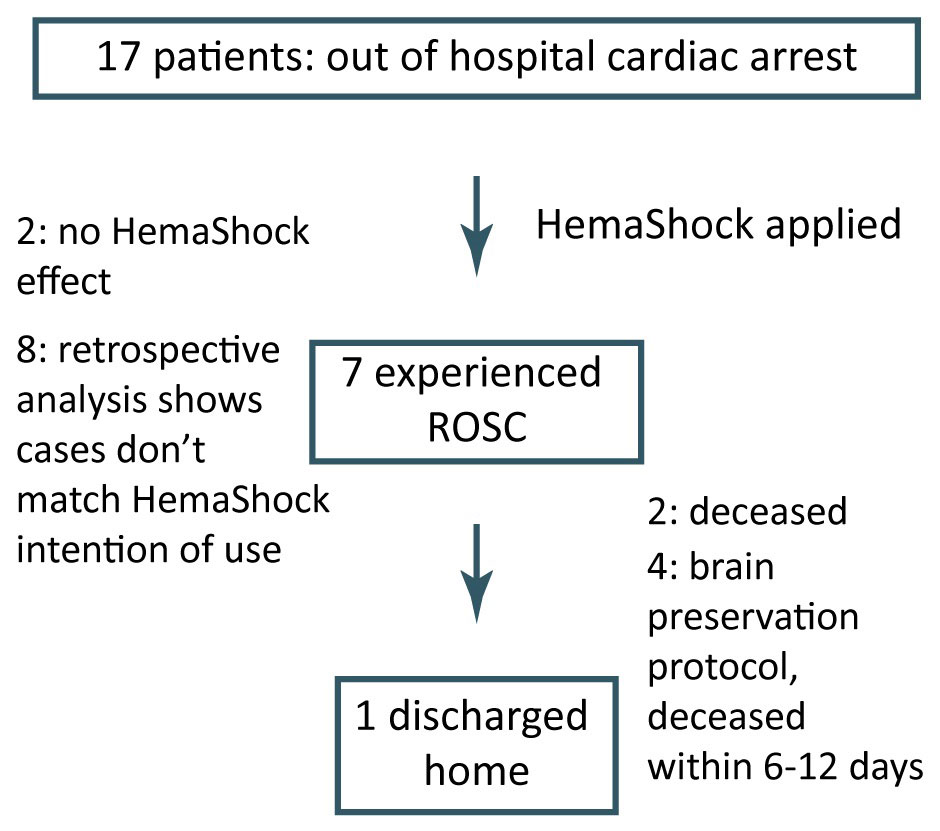
Preliminary clinical validation of the HemaShock® in cardiac arrest cases was carried out at the Eisenhower medical center, Rancho Mirage, CA, USA. HemaShock® was applied to 17 patients, presenting to the ED with cardiac arrest (Figure). All patients presented in critically end-stage status, having failed prehospital efforts including epinephrine, atropine, some amiodarone, and defibrillation, all presenting asystole or PEA rhythms, dilated pupils and essentially no vital signs. Once again, device placement was only carried out within a hospital Emergency Department, with significant time gap from the patients’ collapse to the application of HemaShock®.
7 out of the 17 cases experienced Return of Spontaneous Circulation (ROSC) within 1-5 minutes from HemaShock® application, and 1 patient was eventually discharged home in intact neurological status after brain protection (cooling) treatment in the ICU. Out of the remaining 6, 4 proceeded to brain preservation protocol but decease within 6-12 days, 2 patients were pronounced dead in the ED. No effect of HemaShock® was observed in 10 patients. Retrospective analysis showed that the HemaShock® application was not indicated (e.g. internal bleeding from cancer, extreme hyperthermia etc.).